Therapeutic Products - Events Registration of Interest
Overview
Consultation on an exposure draft of the Therapeutic Products Bill and an associated consultation paper is underway. The Bill is intended to repeal and replace the Medicines Act 1981 and establish a new regulatory scheme for therapeutic products.
Why your views matter
We are holding focus meetings on specific topics to assist stakeholders to provide feedback on the regulatory proposals.
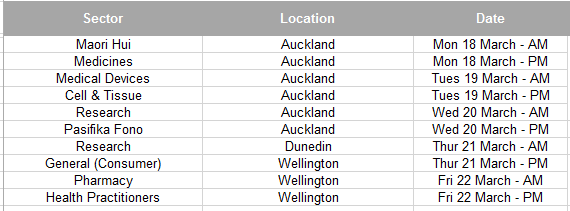
List of focus meetings we are proposing;
What happens next
Once registrations have closed, you will receive an email with event information.
Audiences
- Disability sector
- Gambling sector
- Health sector
- Members of the public
- Mental health and addiction services
- Ministry staff
- People affected by gambling harm
- Pharmaceutical companies
- Service providers
Interests
- Accommodation
- Assisted reproduction
- Cancer services
- Change programme
- Electives
- Emerging Health Technologies
- Gambling
- Health information standards
- Medical devices
- Mental health
- Nutrition and physical activity
- Pacific health
- Pharmacy
- Research
- Screening
- Substance use
- Technology
- Therapeutic products
- Travel

Share
Share on Twitter Share on Facebook